Clinical Trials Market Forecast: Industry Structure, Key Players & Investment Outlook
Clinical Trials Market Size by Phase, Service Type, Indication, Study Design, Region – Segment-Level Market Assessment, Growth Opportunity Analysis, Competitive Mapping & Forecast to 2032
Market Overview
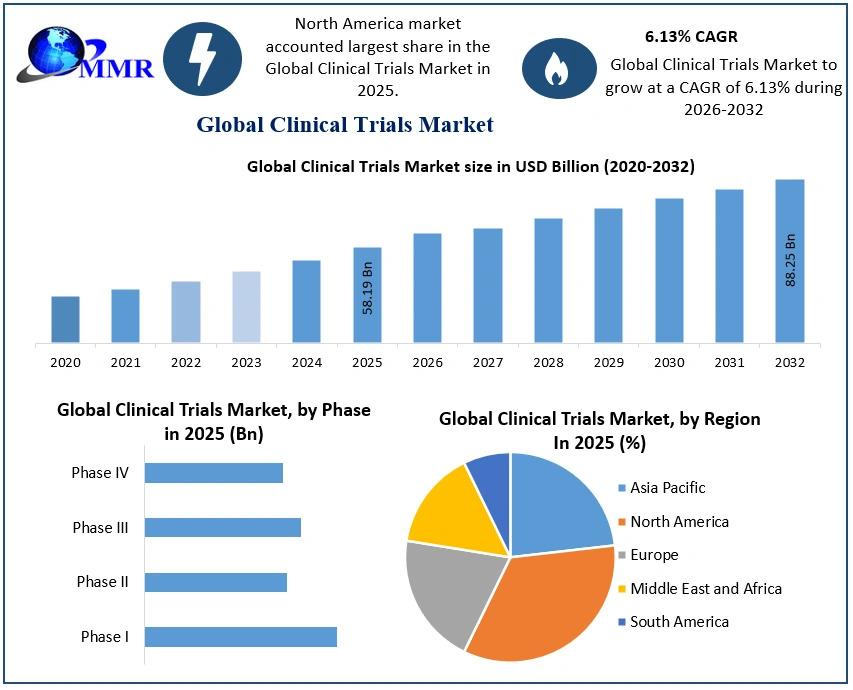
The global Clinical Trials Market was valued at USD 58.19 Billion in 2025 and is projected to reach nearly USD 88.25 Billion by 2032, expanding at a CAGR of 6.13% during 2026–2032. Clinical trials are structured research studies conducted to evaluate the safety, efficacy, and outcomes of new drugs, biologics, medical devices, and therapeutic interventions before regulatory approval. The market encompasses protocol development, patient recruitment, laboratory testing, bioanalytical services, supply chain logistics, decentralized solutions, and data management services.
Rising biopharmaceutical R&D investments, increasing chronic disease prevalence, and growing demand for personalized medicine are key contributors to market expansion. Major contract research organizations (CROs) such as IQVIA Holdings Inc., PPD, Parexel International Corporation, and Charles River Laboratories International, Inc. continue to reshape the competitive landscape through decentralized trial models, AI-enabled analytics, and strategic collaborations.
The acceleration of decentralized and virtual trials—particularly after the COVID-19 pandemic—has transformed patient recruitment, remote monitoring, and data collection methodologies. Regulatory agencies increasingly accept real-world evidence (RWE), enhancing flexibility and improving trial efficiency. These advancements are driving structural shifts in clinical development strategies worldwide.
♦ Request a Free Sample Copy or View Report Summary:https://www.maximizemarketresearch.com/request-sample/189646/
Clinical Trials Market Dynamics
Growth Drivers
1. Rising Demand for Personalized Therapies and Precision Medicine
The growing shift toward targeted therapies and biomarker-driven drug development has intensified the need for well-structured and large-scale clinical trials. Oncology remains the most research-intensive indication area, with thousands of Phase II and Phase III studies underway globally. Personalized medicine requires complex trial designs and advanced laboratory services, boosting overall market demand.
2. Increasing Burden of Chronic and Infectious Diseases
Chronic diseases account for nearly 74% of global deaths, significantly increasing the demand for innovative therapeutics. In the United States alone, over 125 million individuals suffer from chronic conditions, fueling clinical development programs in cardiology, neurology, oncology, and immunology.
3. Decentralized Clinical Trial (DCT) Adoption
The adoption of decentralized models has grown substantially, with an estimated 25% increase in implementation in 2023. Digital tools, wearable devices, telemedicine, and AI-driven patient identification platforms are reducing trial timelines and improving patient retention.
4. AI and Data Analytics Integration
Artificial intelligence and machine learning technologies streamline site selection, patient matching, and predictive analytics, enhancing operational efficiency and reducing overall costs.
Market Restraints
Despite strong growth, the clinical trials market faces challenges:
-
Stringent Regulatory Frameworks: Delays in approvals from agencies such as the FDA and the European Medicines Agency (EMA) can extend timelines and increase costs.
-
High Capital Requirements: Phase III trials require large patient populations and extended durations, significantly increasing operational expenses.
-
Patient Recruitment Challenges: Enrolling diverse and eligible participants remains a persistent obstacle.
-
Data Security Concerns: Growing digitization increases the risk of cybersecurity threats and compliance issues.
Segment-Level Market Assessment
By Phase
Phase III trials dominate the market due to their critical role in confirming safety and efficacy across large populations (up to 3,000 participants). These trials are longer, more complex, and cost-intensive compared to Phase I and II, contributing significantly to revenue share.
Phase I and Phase II trials remain essential for safety and dose-finding studies, while Phase IV post-marketing surveillance is expanding as regulatory agencies demand long-term safety data.
♦ Request a Free Sample Copy or View Report Summary:https://www.maximizemarketresearch.com/request-sample/189646/
By Service Type
Laboratory Services led the market in 2025, given their application across all clinical phases. Bioanalytical testing, biomarker analysis, and central laboratory functions are indispensable for data validation.
Clinical trial supply and logistics services are projected to witness strong growth, driven by globalization of trials and expansion into emerging markets. Decentralized clinical services are also gaining traction due to remote patient engagement capabilities.
Other critical service segments include:
-
Protocol Designing
-
Site Identification
-
Patient Recruitment
-
Clinical Trial Management Services
-
Medical Device Testing Services
By Indication
Oncology remains the largest segment, driven by continuous innovation in immunotherapy, targeted therapy, and cell and gene therapy. Infectious diseases, cardiology, neurology, women’s health, and genetic disorders also contribute significantly.
The increasing pipeline of oncology drugs and personalized cancer treatments supports sustained growth in this segment.
By Study Design
Interventional trials dominate due to their structured methodology and regulatory preference for randomized controlled trials (RCTs).
Observational trials are expanding with growing interest in real-world evidence and post-marketing surveillance. Expanded access trials provide investigational therapies to patients outside traditional trial settings, supporting compassionate use initiatives.
Regional Insights
North America – Market Leader
North America holds the largest share of the global clinical trials market and is expected to maintain dominance through 2032. The United States accounts for over 30% of global trials, supported by advanced infrastructure, high R&D spending, and strong regulatory frameworks.
Canada’s Clinical Trials Fund (CTF), backed by a USD 250 million investment, aims to strengthen trial infrastructure and researcher development. Innovation-driven pharmaceutical companies and CROs further reinforce regional leadership.
Europe
Europe remains a strong contributor with robust regulatory systems and high research intensity in oncology and rare diseases. Countries such as Germany, France, and the UK are key hubs for multinational trials.
Asia-Pacific
Asia-Pacific is the fastest-growing region, driven by cost advantages, large patient pools, and improving regulatory environments in China, India, Japan, and South Korea. CRO expansion in this region supports global outsourcing strategies.
South America and Middle East & Africa
Emerging economies such as Brazil, South Africa, and GCC nations are increasingly participating in global multicenter trials due to regulatory reforms and expanding healthcare infrastructure.
♦ Make Smarter Decisions in 2026 — Get Your Research Report Now: https://www.maximizemarketresearch.com/market-report/clinical-trials-market/189646/
Competitive Landscape & Strategic Mapping
The clinical trials ecosystem is highly competitive, featuring global CROs, regional service providers, and digital health innovators.
Leading companies include:
-
IQVIA Holdings Inc.
-
Parexel International Corporation
-
ICON plc
-
Medpace Holdings, Inc.
-
WuXi AppTec
Tier-I players emphasize decentralized platforms, AI integration, wearable device monitoring, and synthetic control arms. Mid-tier companies focus on niche therapeutic expertise and emerging market penetration.
Strategic partnerships, mergers, digital transformation, and biomarker-driven trial models are shaping competitive positioning across the industry.
◉ Get Market Research Latest Trends
Global Diamond Saw Blade Market https://www.maximizemarketresearch.com/market-report/global-diamond-saw-blade-market/80099/
Global Thin Film Coatings Market https://www.maximizemarketresearch.com/market-report/global-thin-film-coatings-market/102853/
Global Syrup Market https://www.maximizemarketresearch.com/market-report/global-syrup-market/10817/
About Maximize Market Research:
Maximize Market Research is a multifaceted market research and consulting company with professionals from several industries. Some of the industries we cover include medical devices, pharmaceutical manufacturers, science and engineering, electronic components, industrial equipment, technology and communication, cars and automobiles, chemical products and substances, general merchandise, beverages, personal care, and automated systems. To mention a few, we provide market-verified industry estimations, technical trend analysis, crucial market research, strategic advice, competition analysis, production and demand analysis, and client impact studies.
Contact Maximize Market Research:
3rd Floor, Navale IT Park, Phase 2
Pune Bangalore Highway, Narhe,
Pune, Maharashtra 411041, India
sales@maximizemarketresearch.com
+91 96071 95908, +91 9607365656
- Art
- Causes
- Crafts
- Dance
- Drinks
- Film
- Fitness
- Food
- Spiele
- Gardening
- Health
- Startseite
- Literature
- Music
- Networking
- Andere
- Party
- Religion
- Shopping
- Sports
- Theater
- Wellness


